Catalog
-
Catalog
- Agriculture
- Apparel
- Automobiles & Motorcycles
- Beauty & Personal Care
- Business Services
- Chemicals
- Construction & Real Estate
- Consumer Electronics
- Electrical Equipment & Supplies
- Electronic Components & Supplies
- Energy
- Environment
- Excess Inventory
- Fashion Accessories
- Food & Beverage
- Furniture
- Gifts & Crafts
- Hardware
- Health & Medical
- Home & Garden
- Home Appliances
- Lights & Lighting
- Luggage, Bags & Cases
- Machinery, Hardware & Tools
- Measurement & Analysis Instruments
- Mechanical Parts & Fabrication Services
- Minerals & Metallurgy
- Office & School Supplies
- Packaging & Printing
- Rubber & Plastics
- Security & Protection
- Service Equipment
- Shoes & Accessories
- Sports & Entertainment
- Telecommunications
- Textiles & Leather Products
- Timepieces, Jewelry, Eyewear
- Tools
- Toys & Hobbies
- Transportation
Filters
Search
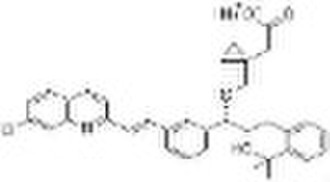
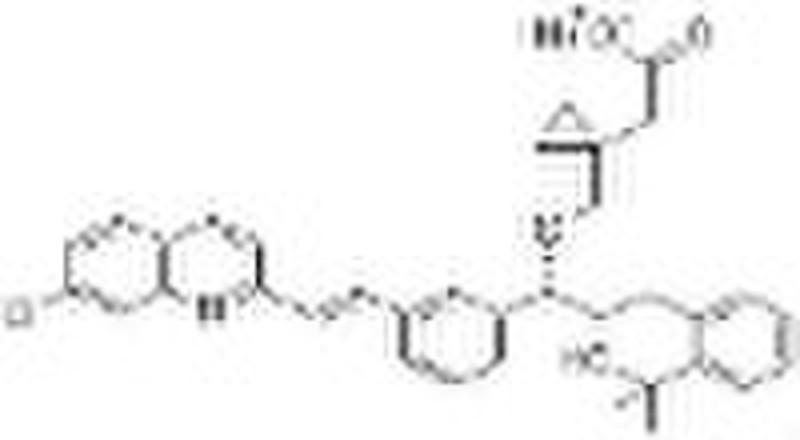
Montelukast Sodium (151767-02-1)
original price: 1,00 USD
Hangzhou, China
Production capacity:
50 Carton / Week

Michael zheng
Contact person
Basic Information
Montelukast can be obtained by two related ways: 1) The Grignard reaction of 3-[2(E)-(7-chloroquinolin-2-yl)vinyl]benzaldehyde (I) with vinylmagnesium bromide (II) in toluene/THF gives the expected secondary alcohol (III), which is condensed with methyl 2-bromobenzoate (IV) by means of palladium acetate and lithium acetate in DMF to yield methyl 2-[3-[3-[2(E)-(7-chloroquinolin-2-yl)vinyl]phenyl]-3-oxopropyl]benzoate (V). The enantioselective reduction of the keto group of (V) with (-)-B-chlorodiisopinocampheylborane in THF affords methyl 2-[3-[3-[2(E)-(7-chloroquinolin-2-yl)vinyl]phenyl]-3(S)-hydroxypropyl] benzoate (VI), which is reacted with methylmagnesium bromide in toluene/THF or methylmagnesium chloride/CeCl3 in THF to give the expected tertiary diol (VII). The selective esterification of (VII) with mesyl chloride and diisopropylethylamine in toluene/acetonitrile yields the expected secondary mesylate (VIII), which is condensed with 2-[1-(sulfanylmethyl)cyclopropyl]acetic acid (IX) by means of butyllithium in THF to afford the corresponding condensation product as free acid that is separated by addition of dicyclohexylamine and precipitates the corresponding salt (X). Finally, this dicyclohexylamine salt (X) is treated with NaOH in toluene/water. 2) The 2-[1-(sulfanylmethyl)cyclopropyl]acetic acid (IX) has been obtained as follows: The reaction of 1,1-cyclopropanedimethanol (XI) with SOCl2 or diisopropyl sulfite in dichloromethane gives 1,1-cyclopropanedimethanol cyclic sulfite (XII), which is treated with NaCN in dichloromethane yielding 2-[1-(hydroxymethyl)cyclopropyl]acetonitrile (XIII). The reaction of (XIII) with mesyl chloride and triethylamine affords the corresponding mesylate (XIV), which is treated with potassium thioacetate in isopropyl acetate giving 2-[1-(acetylsulfanyl)cyclopropyl]acetonitrile (XV). Finally, this compound is hydrolyzed with NaOH in toluene/water to afford (IX).
Delivery terms and packaging
Packaging Detail: 25KG paper drum Delivery Detail: one-two weeks
Port: shanghai/qingdao
Payment term
Documents Against Payment
Letter of credit
Telegraphic transfer
-
Payment Methods
We accept: